Appearance
Metformin
Overview
- Metformin is the first line treatment for type 2 diabetes in most countries. It is cheap, effective at lowering blood glucose and has been shown to reduce the risk of death in people with type 2 diabetes.
- Patients who normally take metformin may benefit from having it continued when they are admitted to ICU as it is effective at controlling blood glucose without the risk of hypoglycaemia and minimises the need for insulin.
- Metformin can cause lactic acidosis in overdose or when there is severe kidney injury as it's only excreted by the kidney, we therefore advise stopping when AKI stage 3 is present, when lactate is > 2mmol/l or when on high doses of vasopressors (ie at risk of lactic acidosis)
- It can be safely restarted once a patient's acute criticall illness is resolving and they can take enteral medications
- For those with hyperglycaemia and a new diagnosis of diabetes or who don't normally take metformin, we consider starting metformin to reduce the need for IV insulin.
- Most metformin is prescribed as an Modified Release preparation (which can't go down a feeding tube). We recommend using liquid solution whn in critical care. This can be prescribed tds.
- We start metformin at 500mg tds but can be increased to 1g bd.
As always, this is for education purposes and not a guideline. Please follow your own unit guidelines, a a link to ours can be found here for reference.
Metformin pharmacology
A lot is known about how metformin works at many different sites in the body but as it's effects are so numerous, a complete description of it's mechanisms of action is yet to be elucidated. For an excellent overview of it's mechanisms of action that have been described see these two articles in Nature Endocrinology Reviews. The first by Foretz et al covers it's glucose lowering effects, the second, again by Foretz et al covers some of it's many non glucose lowering effects, including those acting on the immune system.
Mechanisms of action

Metformin has been used as a herbal remedy for centuries before it's description as a glucose lowering drug in the early 1900's. It is a biguanide derived from the naturally occuring biguanide found in the french lilac plant.
Biguanides are two molecules of guanidine bonded together. The molecule itself is small and is therefore filtered easily by the glomerulus.
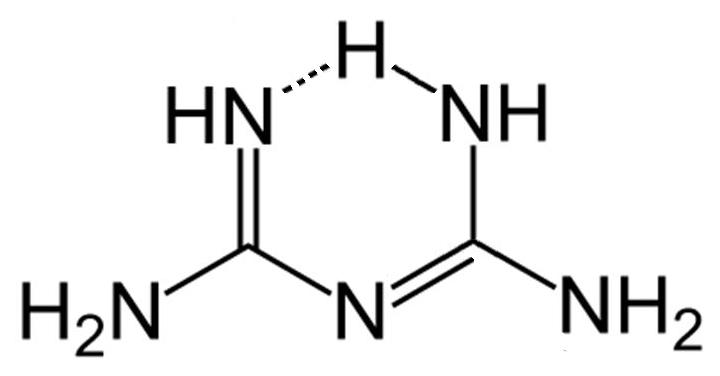
It is so well excreted that it displays "flip-flop" pharmacokinetics which is where the rate of excretion from the body exceeds the rate of absorption. For this reason it's generally given as a modified release to ensure a constant plasma concentration throughout the day.
Glucose lowering effects
Metformin seems to work through multiple pathways
- Reducing hepatic gluconeogenesis - the liver produces out about 200g of glucose a day in normal health. Metformin acts to block the function of the mitocondria within the hepatocyte which reduces the amount of glucose being released into the blood. This is thought to happen through a few different routes including blocking the production of ATP and enzymes that induce gluconeogensis
- In the intestine - metformin has been shown to increase glucose use by enterocytes on the intestinal wall which may reduce the amount of glucose entering the circulation
- The Microbiome - one study showed that metformin that is given in a delayed release tablet and that remained in the gut without being absorbed, showed powerful glucose reducing effects suggesting an interaction with the microbiome.
Non-glucose effects
- As metformin acts on intracellular pathways in many different cell types, it is thought to have effects on immune function, cellular growth and inflammation. This may be important in that there is the suggestion that it may be protective in patients who present with critical illness (see below).
- Metformin has been shown to reduce the risks of cardiovascular disease, cancer and stroke and it is thought that this may be separate to just it's glucose lowering effect in diabetes. It's effect on the enzymes involved in cellular proliferation make it an interesting drug for reducing the risk of these diseases in people who don't have diabetes and it is being trialled as a drug for longevity.
Safety in Critical Care
Most of the concerns about safety for metformin are related to the risk of lactic acidosis. Before reviewing this it's important to state that metformin does not cause hypoglycaemia in normal use and this should not be a consideration when deciding to prescribe. It does cause gastrointestinal side effects in normal use such as diarrhoea and bloating which is a common reason for it to be stopped in the community. This is rarely a contraindication in critical care
Metformin Associated Lactic Acidosis
This is a serious and not entirely rare complication of metformin use. Metformin is not metabolised by the liver and is excreted unchanged in the urine. It is a small molecule so filtered easily and renal function needs to decline significantly before toxic effects are apparent. Most cases of MALA are in patients who continue to take metformin and experience an acute illness that causes renal failure. The presentation is dramatic with severe lactic acidosis and whilst it is quickly reversible with resuscitation and renal replacement, mortality remains high.
Traditionally metformin has been held in patinets admitted to intensive care either (rightly) because of the concern about lactic acidosis or (perhaps wrongly) because of concerns about hypoglycaemia.
Lactate is certainly higher in patients who present to ICU and who are taking metformin and is significantly higher still if they have an acute kidney injury, as shown in this study by Postma et al in 2020.
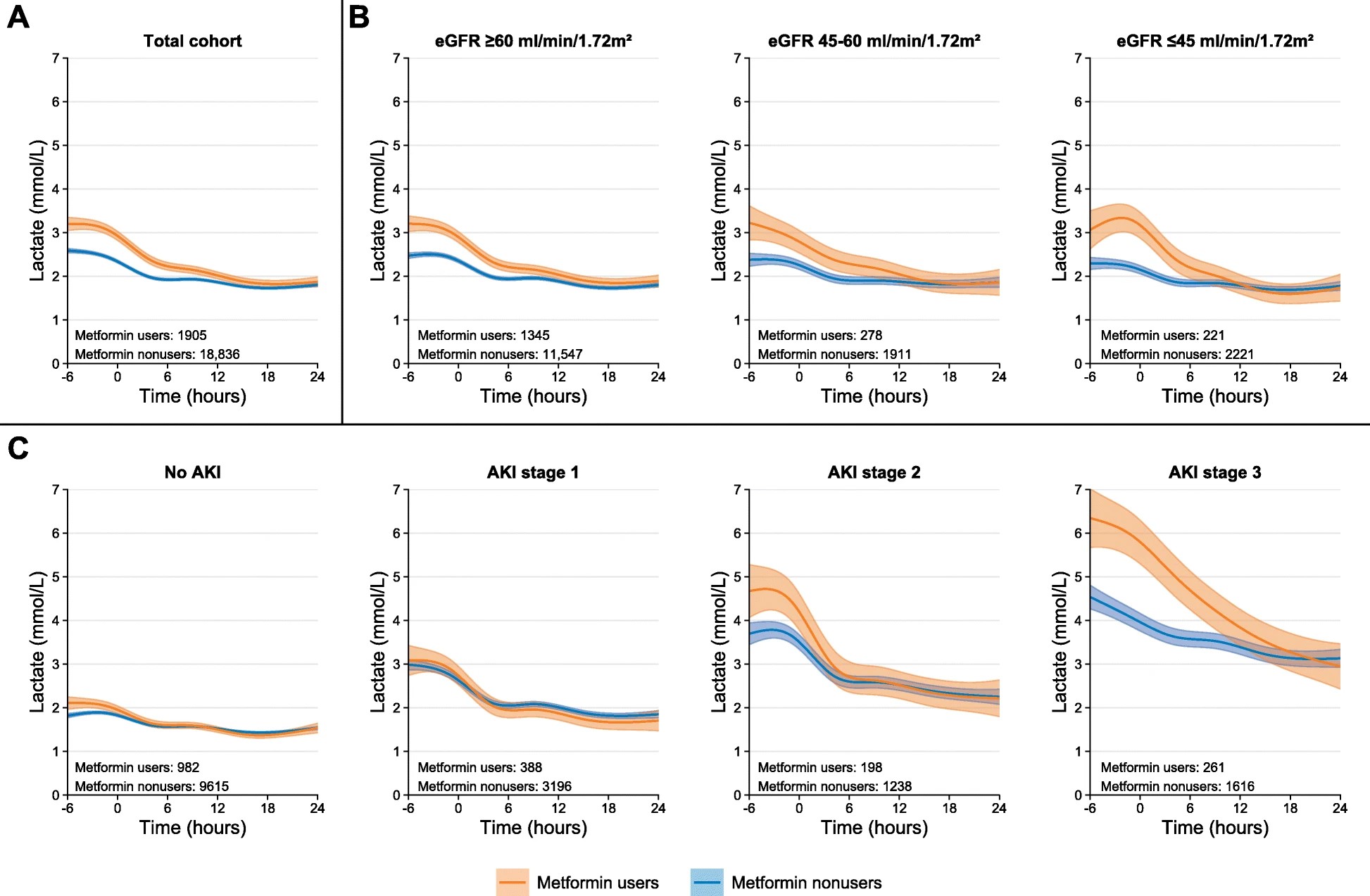
Image reproduced from Postma et al.[1] Mean lactate trajectories over time with 95% confidence interval for metformin users and nonusers were fitted by a mixed-effect model with individual-level random intercept and slope. Time after ICU admission was modeled as natural cubic spline with knot location at − 1 h, + 4 h, and + 12 h surrounding intensive care unit admission. a Total population. Subsequently, analyses were stratified according to b chronic kidney disease stage based on mean estimated glomerular filtration rate (eGFR) 1 year before ICU admission or c acute kidney injury (AKI) stage within 24 h of ICU admission
Metformin associated lactic acidosis is a serious complication that can arise in overdose or in patients who develop acute kidney injury and continue to take metformin
In the absence of end stage renal failure and lactic acidosis, is metformin safe for use in critical care?
There are as yet no randomised control studies to answer this question but there are plenty of retrospective cohort studies that show an interesting association between metformin use and better outcomes from critical illness. This has been shown in influenza, COVID-19 and in cohorts of all causes presenting to ICU.
A retrospective propensity matched cohort study from Gomez et al looked at 14,847 patients in the US from 2008-2014 presenting with sepsis and a history of diabetes mellitus. They matched patients who had exposure to metformin during their hospitlisation with controls and found a decreaed 90-day mortality (11.9% vs 22.7%, OR 0.46). They concluded that "Metformin exposure during hospitalization is associated with a decrease in 90-day mortality in patients with type 2 diabetes mellitus and sepsis."
An earlier study done in Denmark by Christansen et al[2] looked at patients with type 2 diabetes admitted to intensive care between 2005-11 and compared metformin use before admission those without metformin exposure. Mortality was significantly different (17.6% in the metformin group, 25% in non-metformin users. The authors concluded that "Preadmission metformin use was associated with reduced 30-day mortality among medical and surgical intensive care patients with type 2 diabetes."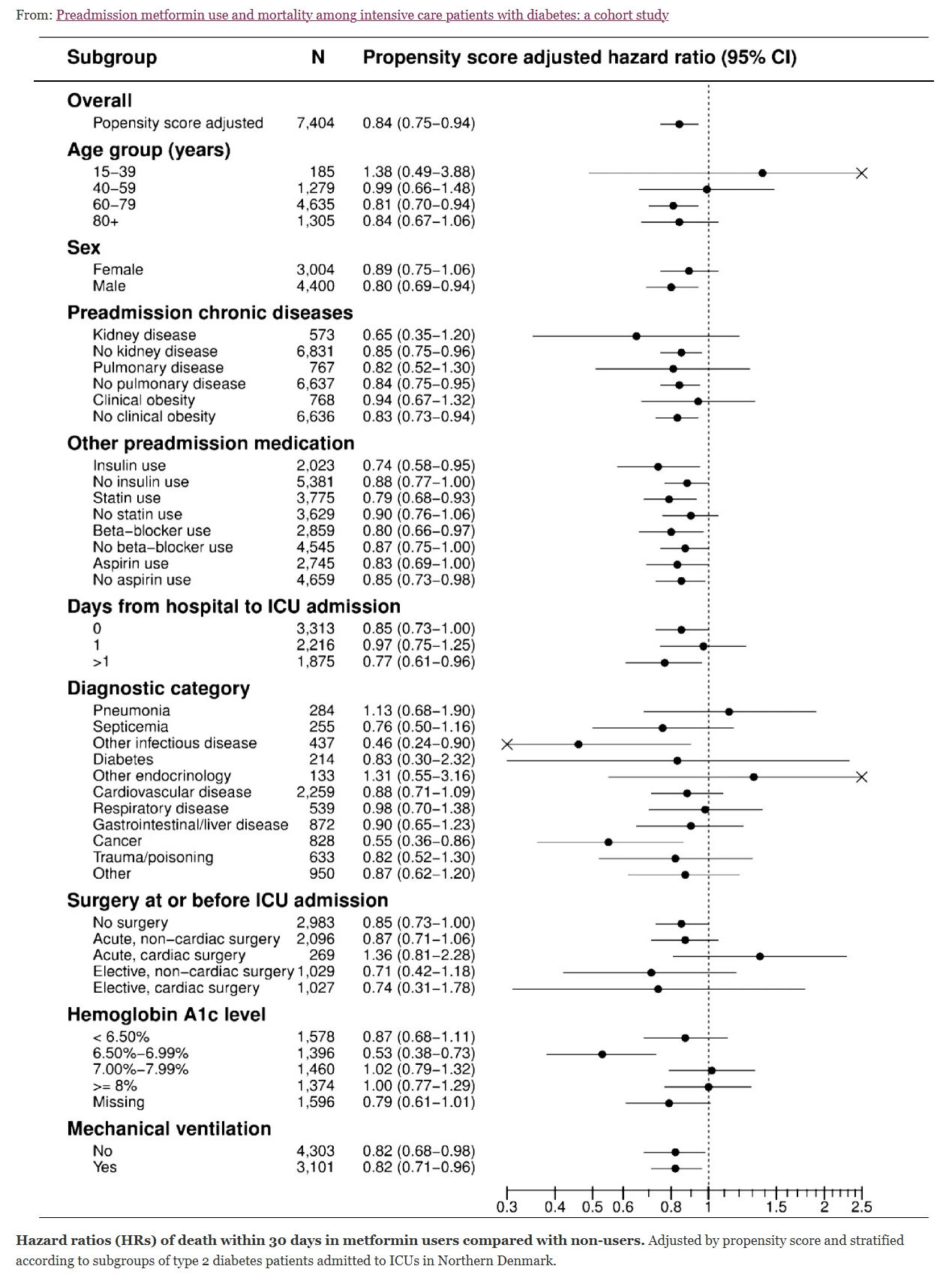
There is evidence that metformin exposure before and during admission to critical care is associated with a reduced risk of mortality
How can we use it safely?
Our experience using metformin in critical care is that it is effective at lowering blood glucose and may reduce the need for treatment with insulin infusions.
We stop metformin in anyone who has stage 3 acute kidney injury, lactic acidosis or who is at risk of lactic acidosis (shock requiring large doses of vasopressors).
In the absence of these contraindications we start metformin in any patient who usually takes it pre-admission and start it in any patient who is known type 2 diabetic or who is diagnosed with diabetes on admission (this is why checking HbA1c on admission and getting the right diagnosis is so important). Most patients who take metformin are prescribed a modified release preparation which cannot be given down an enteral feeding tube. For this reason we use a liquid preparation. It can be given tds to make it's effect last across the day and the usual dose can be prescribed. When starting it for patients not previously taking it we start at 500mg tds but this can be increased to a maximum of 1g bd.
Posma, R.A., Hulman, A., Thomsen, R.W. et al. Metformin use and early lactate levels in critically ill patients according to chronic and acute renal impairment. Crit Care 24, 585 (2020). https://doi.org/10.1186/s13054-020-03300-y ↩︎
Christiansen et al.: Preadmission metformin use and mortality among intensive care patients with diabetes: a cohort study. Critical Care 20132013 17:R192 doi:10.1186/cc12886 ↩︎